選択した画像 ”¯Œ^ ƒƒ“ƒO 40‘ã ƒXƒgƒŒ[ƒg 119581
300 g O 2 1 mol O x 2 3 g O 2 2 1 mol CO x 2 mol O 2 = 0469 mol CO The second assumption is correct Therefore, oxygen is the limiting reactant 2 2 440 g 0469 mol CO x = 6 g CO 1 mol 12 In an experiment with Zn and S, it was found that 307 g of ZnS was produced If the(16 kJ)/(05 mole H 2 (g)) = 3672 kJ per mole of H 2 (g);400 g C are 671 g H 533 g O mol C 11 g C mol H 1008 g H mol O 1600 g O = 333 mol C = 666 mol H = 333 mol O CH 2O empirical formula mass of CH 2O molar mass of lactate 9008 g 3003 g 3 C 3H 6O 3 is the molecular formula O 333 333 H 666 333 C 333 333 Combustion apparatus for determining formulas of organic compounds Figure 35

Variation Of Dt X 2 T G Th Vs X Mol Of Pbo Content In A D90 2 Xthsb 2 Download Scientific Diagram
"¯Œ^ ƒƒ"ƒO 40'ã ƒXƒgƒŒ[ƒg
"¯Œ^ ƒƒ"ƒO 40'ã ƒXƒgƒŒ[ƒg-O(Ans 600 g) (h) A mass of g of "KHP" (potassium hydrogen phthalate;Mass = (425 g x 0133 J/g deg x 754 o) / (418 J/g deg x 25 o) = 408 g mass = _41 g_____ 4 A chemical compound has a molecular weight of 05g/mole 1400 g of this compound underwent complete combustion under constant pressure in a calorimeter with a heat capacity of 2980 J o C1 The temperature went up by 1195 degrees Calculate




Stanley Coffret Outils 38 Pieces Cdiscount Bricolage
About Press Copyright Contact us Creators Advertise Developers Terms Privacy Policy & Safety How works Test new features Press Copyright Contact us CreatorsThe mole ratio of CHO in 100 g of ketonox is given by, € Moles C = 4615 g C 1 mol C 11 g C = 3843 mol C Moles H = 7746 g H 1 mol H 1008 g H = 7685 mol H Moles O = 4611 g C 1 mol O 1600 g O = 28 mol OStep 1 O 3 (g) NO 2 (g) NO 3 (g) O 2 (g) step 2 NO 3 (g) NO 2 (g) N2O5 (g) The experimental rate law is rate = kO 3NO 2 Because the rate law conforms to the molecularity of the first step, that must be the ratedetermining step The second step must be much faster than the first one The decomposition of Hydrogen Peroxide 2 H 2O2 (aq
2N2O5(g)→4NO2(g) O2(g) From the graph looking at t = 300 to 400 s 61 2 M Rate O = 9 10 Ms 540 550 560 570 0 50 100 150 0 250 300 time (s) ln P k = 26x103s1 Reaction HalfLife 12 Reaction Halflife Halflife is the time taken for the concentration ofA 15 kg block of Ni at 100 °C is placed into 500 mL of water that has a temperature of 21 °C What is the final temperature assuming the specific heat of Ni is 044 J/g°C and the specific heat of water is 4184 J/g°CCalculate the number of moles of O (atomic oxygen) in 400 grams of oxygen a) 250 moles of O b) 125 moles of O How many atoms of carbon are present in 345 g of caffeine, C 8 H 10 N 4 O 2?
G t2 (a) we solve for y = h which yields h = 518 m for y 0 = 0, v 0 = 4 m/s, q 0 = 600° and t = 550 s − 0 0y − 2 (b) The horizontal motion is steady, so v x = v 0x = v 0 cos θ 0, but the vertical component of velocity varies according the equations before Thus, the speedi id at impact is v = ()v 0 cosθ 0 2 v 0 sinθ 0 −gt 2742 x 1023 atoms O x (1 mol / 6022 x 1023) = 1232 mol O / 308 = 4 K2MnO4 – potassium manganate 2 Samples of a compound are found to be 2791% Fe, 2408% S, and 4801% O Can you figure out the empirical formula of the anion based on your knowledge of nomenclature?N2(g) 3H2(g) ⇔2NH3(g) ∆Go = 330 kJ K298 = 60 x 105 Thermodynamically favored at 298 K However, rate is slow at 298K Commercial production of NH3 is carried out at temperatures of 800 to 900 K, because the rate is faster even though K is




Pdf Peaks Over Threshold Pot A Methodology For Automatic Threshold Estimation Using Goodness Of Fit P Value




Romertopf 140 05 Plat A Four Terre Cuite 4 A 6 Personnes 3 5 Litres Amazon Fr Cuisine Et Maison
Œ Q)Ô 0\ "ë 'Û‚s„ã0¼ 2‹1* Ÿ 9kSdb©•t&ñ _¡áƒP¹ä OˆÄ ìJ' Nòá9¯¤^" nG ý4ìôÓ@à±Î òn÷‡ý\ÌÀh‰ÖK& 0ãD&y³ØüŠ€ ÁbÉÆ\Jž ´#˜Ècoì k ð_¿8„L2‹Õæ×ÁéJd ˆ ±qª ~qØ(#h 6ï @Ÿ /R ªhøPp˜h)õ\¨)äÏšuÐŒp`3&Ð I¢ Sû¶bj¥¨À lÜð§ÚÂ9³ÅX3"Ã% ' µ "äSearch the world's information, including webpages, images, videos and more Google has many special features to help you find exactly what you're looking forHow many moles are there in 402 x 105 grams of H 2 O?




Pdf Representation Of Numerical Data By Modified Exponential Curve




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr
Mass of a compound of a solution calculation Mass (g) = Concentration (mol/L) x Volume (L) x Molecular Weight (g/mol) The key here is to realize that you're dealing with a hydrocarbon, that is, a compound that contains only carbon and hydrogen Notice that the products of this combustion reaction are carbon dioxide, #"CO"_2#, and water, #"H"_2"O"# This tells you that all the carbon that was initially a part of the hydrocarbon will now be part of the carbon dioxide Likewise, all the hydrogen thatStarting with initial concentrations of 0040 mol/L of N 2 and 0040 mol/L of O 2, calculate the equilibrium concentration of NO in mol/L (a) mol/L (b) mol/L (c) 0011 mol/L (d) 0080 mol/L (e) 010 mol/L 9 K c = 0040 for the system below at 450 o C If a reaction is initiated with 040 mole of Cl 2 and 040 mole of PCl 3 in a liter container, what is the equilibrium




8 5 G High Resolution Stock Photography And Images Alamy



Aip Scitation Org
1008 g H x 1 mol H O 2 mol H x 1802 g H O 1 mol H O 158 x 10 g H O x 2 2 2 23 = 1767 x 104g H The mass percentages of C and H can be calculated using the masses from the previous calculations Percent C = 387 mg 15 mg x 100% = 4090 = 409% C Percent H = 387 mg mg x 100% = = 457% H"to" 1 mol H 2 O "from" 1802 g H 2 O (g of H 2 O) 1 mol H 2 O = 101 mol H 2 O 1802 g H 2 O (00 mol H 2 O) 1802 g H 2 O = 3604 g H 2 O 1 mol H 2 O (4Molar mass of K = 391 g Molar mass of Mn = 549 g Molar mass of O = 160 g Molar mass of KMnO4 = 391 g 549 g (160 g x 4) Molar mass of KMnO4 = 1580 g What other calculations you can do with the molarity calculator?




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr



Jencam De
Q = m x C x DT q = 250g x 418J/g o C x 26 o C q = 37,6J or 38kJ 2 Calculate the specific heat capacity of copper given that 475 J of energy raises the temperature of 15g of copper from 25 o to 60 o q = m x C x DT C= q/m x DT C = 475J /(15g x 35 o C ) C= 039 J/g o C Type your answer here 40g means 40grams Wiki User ∙ 1931 This answer is979 g / 48 g > The moles of O in 306 g of M 2 O 3 48 g > There are 48 g in three moles of oxygen The three comes from oxygen's subscript in the formula (I suppose I should write 48 g/3mol 2x 48 > The molar mas of M 2 O 3 x is the atomic weight of M, note that is multiplied by two because of the subscripted two in the formula




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr




Specific Fuel Consumption Vs Engine Speed Download Scientific Diagram
400 g NaOH Take either of these moles of reactants and calculate how much of the other reactant would be needed to react completely with it Let us take mol FeCl 3 3 mol NaOH (F) mol FeCl 3 x = 0185 mol NaOH needed 1 mol FeCl 3 Compare the moles needed (F) with the available moles from (E), aboveA) 857 x 10 23 b) 268 x 10 25 c) 108 x 10 24 d) 9 x 10 23 e) 4 x 10 23 16 Suppose you have a 100gram sample of each of the following240 mol I x x mol I 1 m 2 3 652 g Al o I 2 l AlI 2 = Now assume I 2 is limiting 11 2Al 3 I2> 2 AlI 3 Solution – Cont (b) Determine the yield of the AlI 3 if one starts with 1 g Al and 240 g I2 21 3 3 3 3 2 2 2 2 1 mol I mol AlI g AlI 240 g I x x x g I 2



Pubs Rsc Org



Hal Sorbonne Universite Fr
Equilibrium Calculations II CHM 152 1 At a particular temperature, Kc = × 10 −6 for the reaction 2 CO2 (g) U 2 CO (g) O2 (g) If mol CO2 is initially placed into a 50 L vessel, calculate the equilibrium concentrations of all species 2 CO2 (g) U 2 CO (g) O2 (g) Initial (M) 040 0 0 Change (M) −2x 2x xEquil (M) 040 − 2x 2x x2 2 c 2 2(414 g O/114 g) x 100 = 363% O with compounds, find molar mass of the compound and molar mass of the element you are trying to find and times by the subscript for that element within the formula, divide molar mass of element by total molar mass of compound and multiply by 100 to find1 Ca = 1 C = 111 3 O = 3 x Molar mass of CaCO 3 = g Iron(II) sulfate, FeSO 4 Molar mass of FeSO 4 = g Chapter 3 Solutions Solution a homogenous mixture in which the components are evenly distributed in each other Solute the component of a




Sandisk Extreme Pro 128 Go Cle Usb 3 1 Gen 1 Ssd Avec Vitesses De Lecture Jusqu A 4 Mo S Et Vitesses D Ecriture Jusqu A 380 Mo S Amazon Fr Informatique
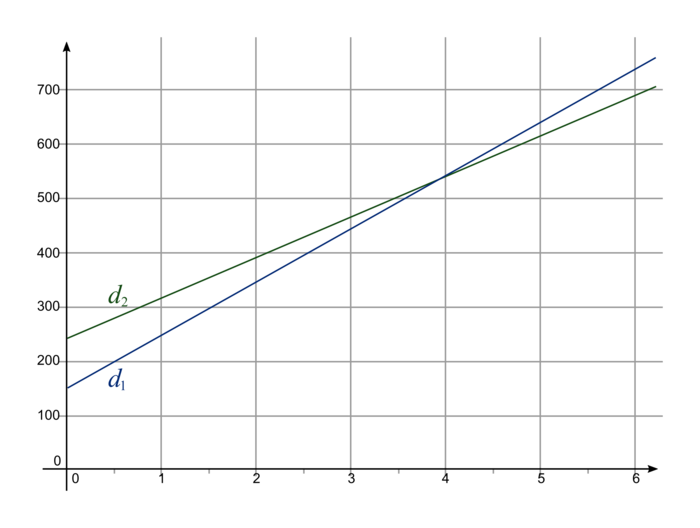



Sujet 19 Groupement Academique 2 Capconcours Cc
321 x 1024 H atoms ∣ 1 mol H ∣ 1 mol H2O ∣ 180 g H2O ∣ = 480 g H2O ∣ 602 x 1023 H atoms ∣ 2 mol H ∣ 1 mol H2O∣ 16 Calculate the number of C atoms in 2568 g of C10H8O 2568 g C10H8O ∣ 1 mol C10H8O ∣ 10 mol C ∣6022 x 10 23 C atoms ∣ =40 101 101 101 101 101 8 80 Bishop Redding San Francisco Eureka Fresno Susanville Ukiah Bakersfield Needles San Luis Obispo El Centro Nevada City D i e g o R i v e r r S u san Rive r S t a n i sl a u River C o s u m n e s R i v e r K n g s R i v e r M o k e l u m n e Riv e r Tuol u m n eRiv r A l a m o R i ve r Fr e s n o Riv e r Y u b a R2C 3 H 6 2NH 3 3O 2 2C 3 3 N 6H 2 O a 368 g b 735 g c 1470 g d 462 g e
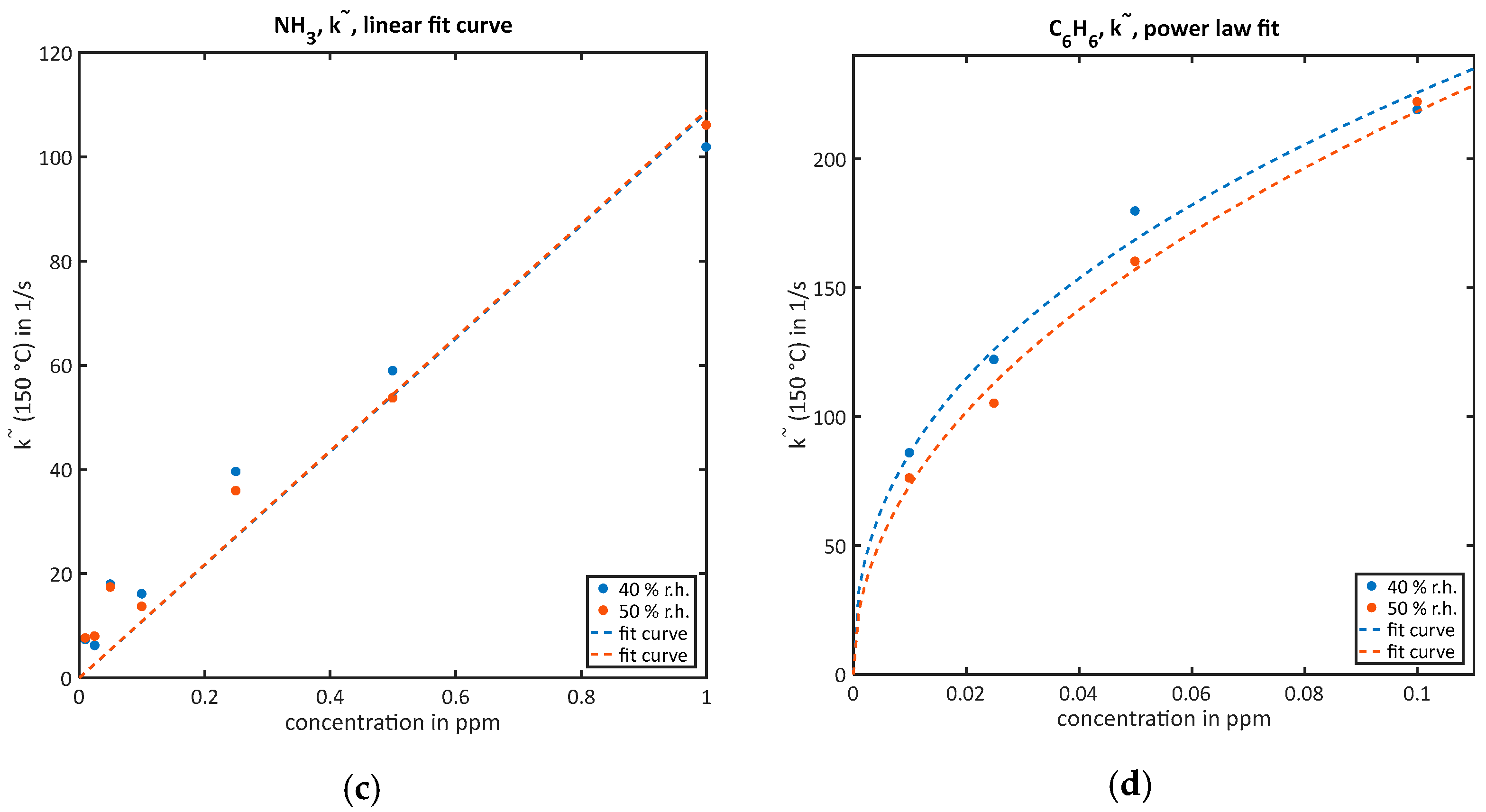



Sensors Free Full Text Facile Quantification And Identification Techniques For Reducing Gases Over A Wide Concentration Range Using A Mos Sensor In Temperature Cycled Operation Html
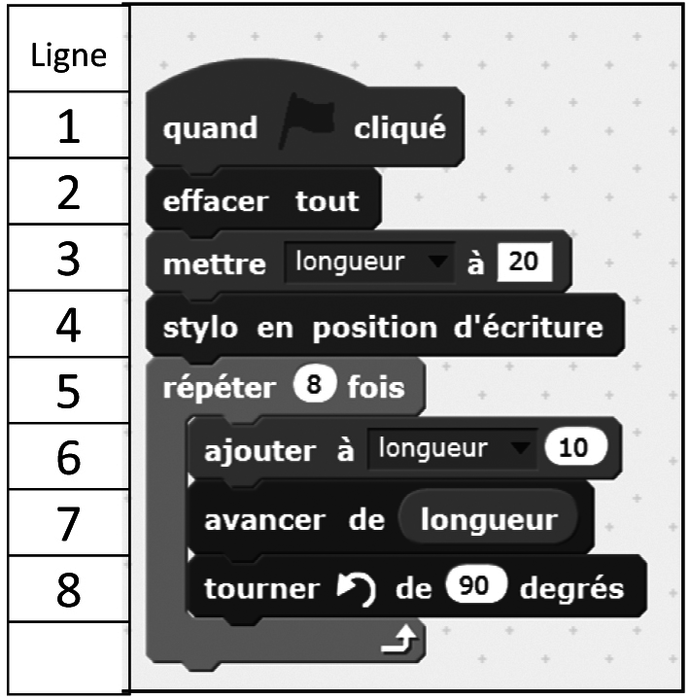



Sujet 19 Groupement Academique 2 Capconcours Cc
Find local businesses, view maps and get driving directions in Google Maps Mass of water = 25 g, Mass of ethyl alcohol = 25 g and mass of acetic acid = 50 g Molecular mass of water (H 2 O) = 1 g x 2 16 g x 1 = 18 g mol1 Molecular mass of ethyl alcohol (C 2 H 5 OH) = 12 g x 2 1 g x 6 16g x 1 = 46 g mol1 Molecular mass of acetic acid (CH 3 COOH) = 12 g x 2 1 g x 4 16g x 2 = 60 g mol142 g/mole) was dissolved in water and made up to 1000 mL in a volumetric flask 1500 mL of the "KHP" solution required 2485 mL




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr




Gs 40 48 D 0 480v 40a Relais Statique Gefran 40 A Montage Panneau 32 V C C 530 V C A Rs Components
2791 g Fe x (1 mol / g) = / = 1 x 2 = 2Find (f g)(x) for f and g below f(x) = 3x 4 (6) g(x) = x2 1 x (7) When composing functions we always read from right to left So, rst, we will plug x into g (which is already done) and then g into f What this means, is that wherever we see an x in f we will plug in g That is, g acts as our new variable and we have f(g(x))Graph g (x)=x g(x) = x g ( x) = x Rewrite the function as an equation y = x y = x Use the slopeintercept form to find the slope and yintercept Tap for more steps The slopeintercept form is y = m x b y = m x b, where m m is the slope and b b is the yintercept y = m x b y = m x b Find the values of m m and b b using the form



Anses Fr




Xebnaiztbrhpzm
Consider as another example a sample of compound determined to contain 531 g Cl and 840 g O Following the same approach yields a tentative empirical formula of Cl 0150 O 0525 = Cl 0150 0150 O 0525 0150 = ClO 35 Cl 0150 O 0525 = Cl 0150 0150 O 0525 0150 = ClO 35O a 400 g b 160 g c 800 g d 640 g e 160 g ____ 27 Acrylonitrile, C 3 H 3 N, is a molecule used to produce a plastic called Orlon How many grams of acrylonitrile could be produced by reacting 5 g of propene, C 3 H 6 with excess ammonia, NH 3 and oxygen?40 A saturated solution of NaNO 3 is prepared at 60°C using 100 grams of water As this solution is cooled to 10°C, NaNO 3 precipitates (settles) out of the solution The resulting solution is saturated Approximately how many grams of NaNO 3 settled out of the original solution?




Sandisk Extreme Pro 128 Go Cle Usb 3 1 Gen 1 Ssd Avec Vitesses De Lecture Jusqu A 4 Mo S Et Vitesses D Ecriture Jusqu A 380 Mo S Amazon Fr Informatique




Veet 40 Bandes De Cire Froides Cdiscount Electromenager
12 g iron / 100 g sample = x g iron / 250 g sample Crossmultiply and divide x= (12 x 250) / 100 = 30 grams of iron How to Calculate Volume Percent Concentration of a Solution Volume percent is the volume of solute per volume of solution This unit is used when mixing together volumes of two solutions to prepare a new solution25 g of aluminum 270 g/mL 1) Plan Calculate the volume for each metal and select the metal sample with the greatest volume 1) 25g x 1 mL = 93 mL aluminum 270 g 2) 45 g x 1 mL = 23 mL gold 193 g 3) 75 g x 1 mL = 66 mL lead 113 g Ref Timberlake, "Chemistry", Pearson/Benjamin Cummings, 06, 9th EdDetermine the masses of each element assuming 100 g 400 g C 671 g H 5329 g O Convert the masses into moles of each element 400 g C x 1 mol C = 333 mol C 11 g C 671 g Hx 1 mol H = 666 mol H 1008 g H 5329 g Ox 1 mol O = 333 mol O 1600 g O Express the moles as the smallest possible ratio CH 2O
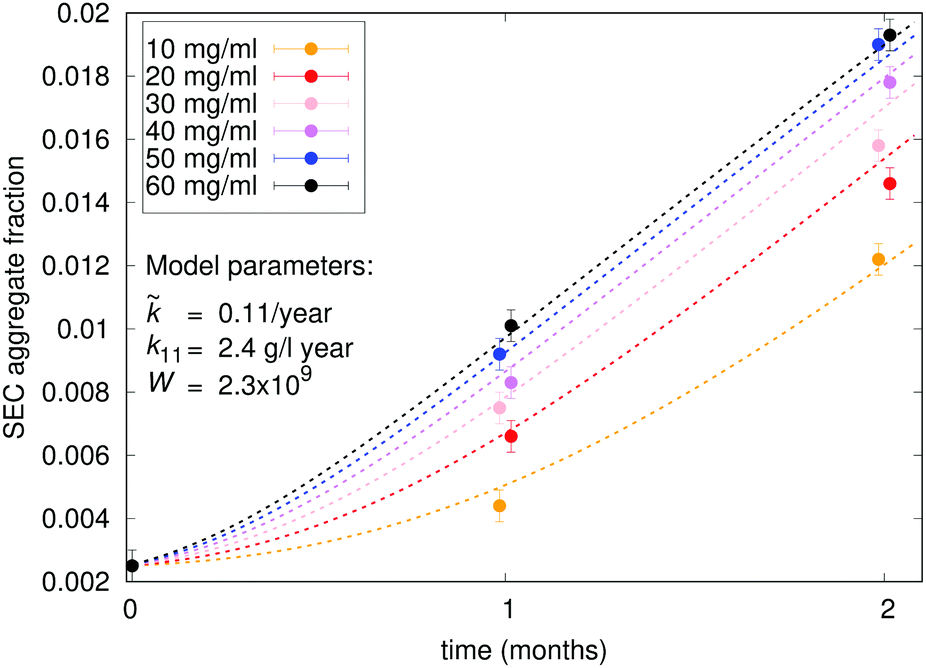



Characterisation Of Protein Aggregation With The Smoluchowski Coagulation Approach For Use In Biopharmaceuticals Soft Matter Rsc Publishing Doi 10 1039 C8smh




Maison Neue Milieu Grotesque Digital Typefoundry
Chain of equalities 1802 g H 2 O = 1 mole of H 2 O The conversion factor How many grams are there in 00 moles of H 2 O?Example 2N2O5(g) →4NO2(g) O2(g) N 2 O 5 c d t b Concentration versus time from experiment a Rate b c d Rate = kN2O5 a N2O5 Experimental rate law k is a constant (slope Slope of a of line) Slope of b Slope of c Slope of d – For most reactions of the typeSkill 35 Lactic acid (M = 9008 g/mol) contains 400 mass % C, 671 mass % H, and 533 mass % O (a) Determine the empirical formula of lactic acid (b) Determine the molecular formula Plan Assume 100 g lactic acid to express each mass % as grams Convert grams to moles and find the empirical formula



Pubs Rsc Org



Hal Archives Ouvertes Fr
(1) 46 g (2) 61 g (3) 85 g (4) 126 g
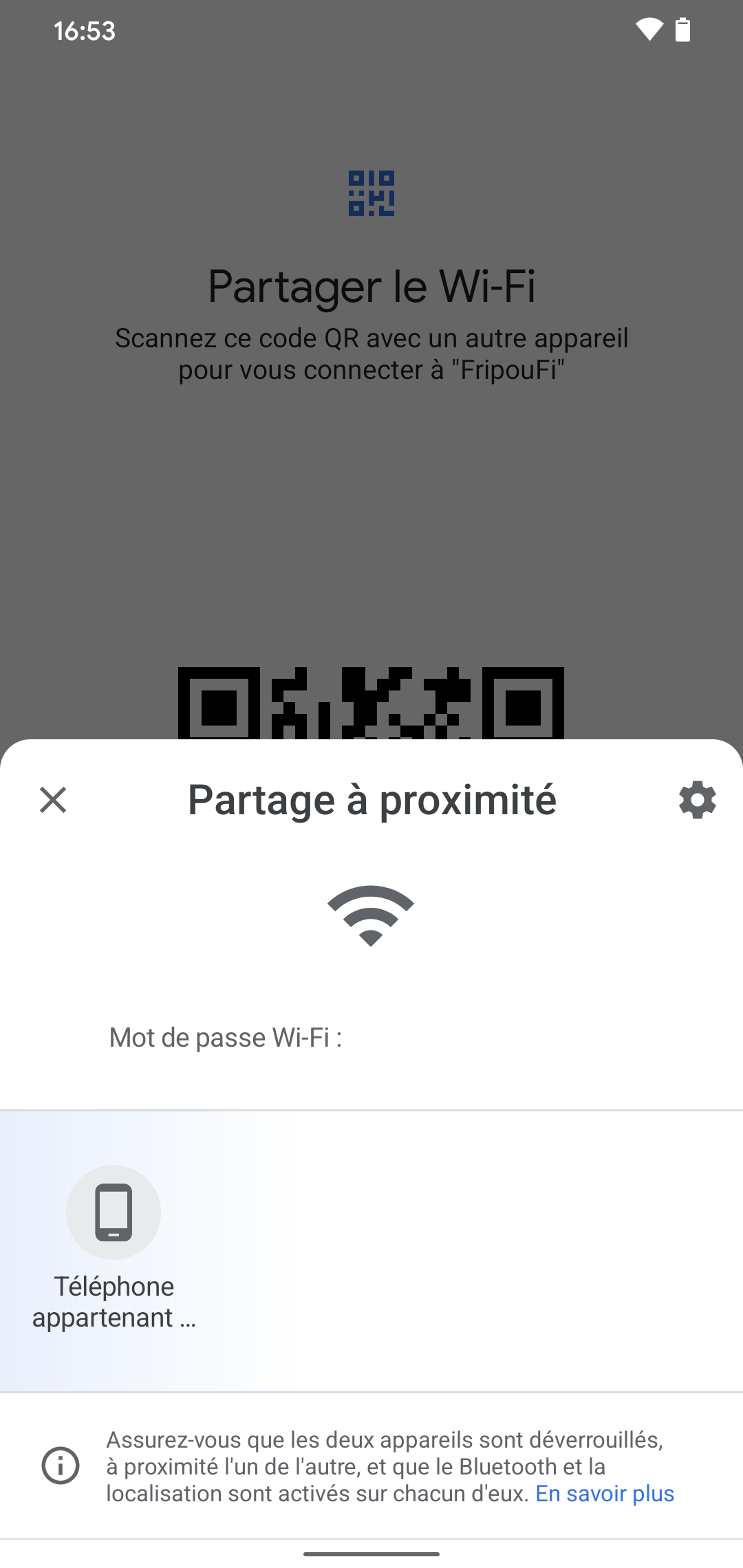



Images Frandroid Com Wp Content Uploads 21 04 Android 12 Partage Wifi 2 Png




Calor Pro Express Ultimate Pure Centrale Vapeur Technologie Professionelle Haute Pression Fonction Pressing Jusqu A 650 G Min Gv9610c0 Amazon Fr Cuisine Et Maison




Pdf Improving Photosynthesis



Leesu Fr



John Libbey Eurotext Bulletin Du Cancer Role Des Lymphocytes T Regulateurs Dans La Progression De La Fibrose Hepatique Associee A L Infection Par Le Vhc




Gate Tunable Bandgap Renormalization And Exciton Binding Energy Of Download Scientific Diagram



Gemu Group Com




Stanley Coffret Outils 38 Pieces Cdiscount Bricolage
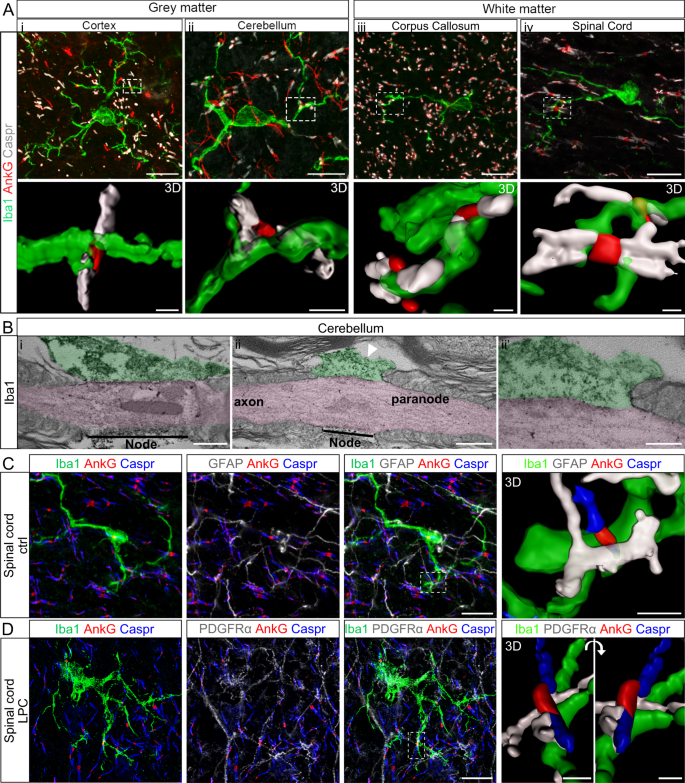



Microglia Neuron Interaction At Nodes Of Ranvier Depends On Neuronal Activity Through Potassium Release And Contributes To Remyelination Nature Communications




Eaton 1673 Disjoncteur Fi 40 A 4 Poles Type G B Chez Reichelt Elektronik
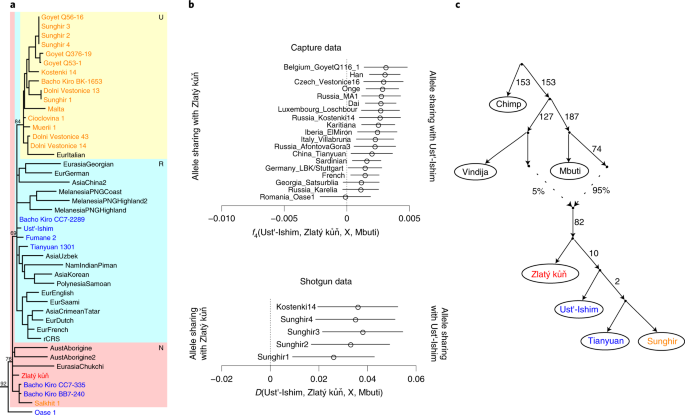



A Genome Sequence From A Modern Human Skull Over 45 000 Years Old From Zlaty Kun In Czechia Nature Ecology Evolution




Serrures A Mortaiser Comparez Les Prix Pour Professionnels Sur Hellopro Fr Page 1
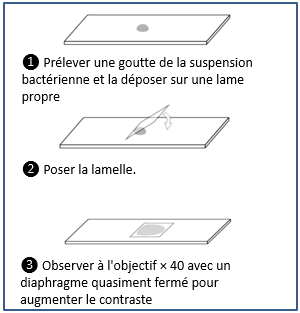



Examen Microscopique En Microbiologie Medicale




M Asam Magic Finish Fond De Teint Mousse Summer Teint Spf 30 Asambeauty France
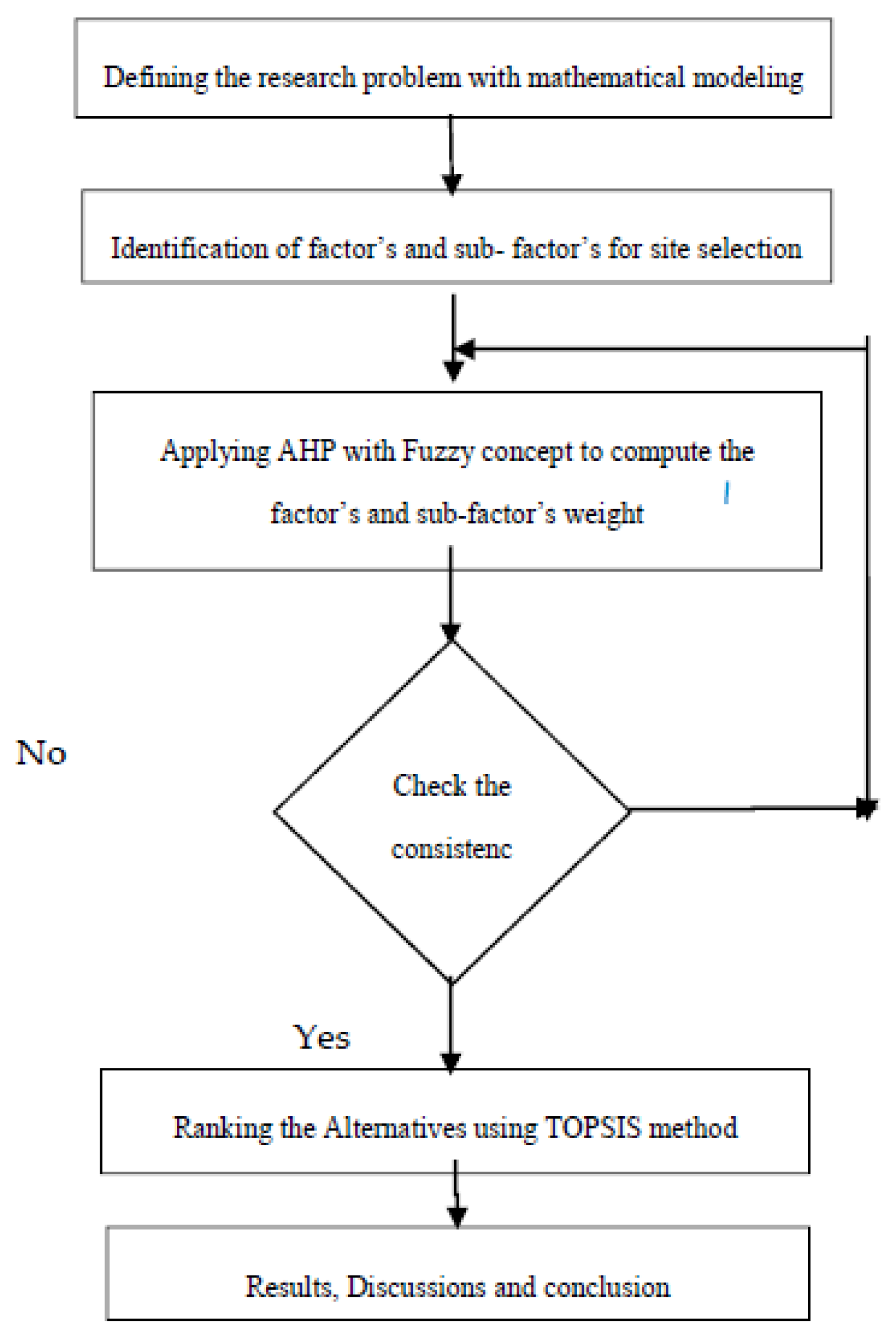



Mathematics Free Full Text Ahp Topsis Inspired Shopping Mall Site Selection Problem With Fuzzy Data Html




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr
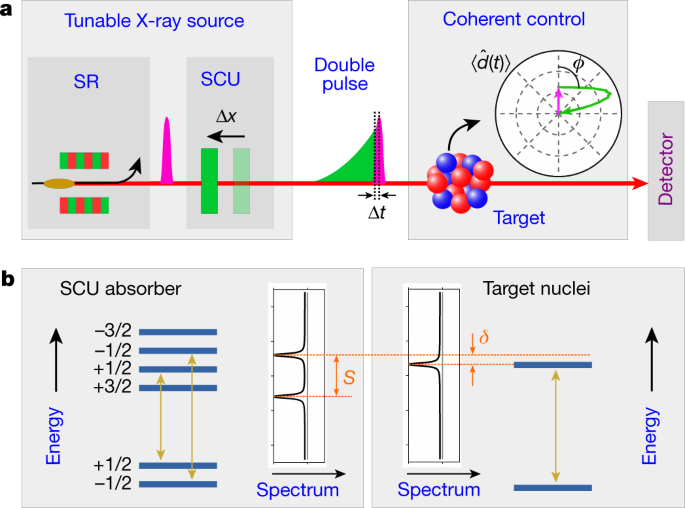



Coherent X Ray Optical Control Of Nuclear Excitons Nature




Google Workspace Ex G Suite La Suite De Productivite De Google




Pdf Effect Of Low Salinity On Microbial Floc Composition And Performance Of Litopenaeus Vannamei Boone Juveniles Reared In A Zero Water Exchange Super Intensive System




My Publications 40 Farameen E Mustafa Page 78 79 Created With Publitas Com




Variation Of Dt X 2 T G Th Vs X Mol Of Pbo Content In A D90 2 Xthsb 2 Download Scientific Diagram



Atlantic Pros Fr




642 Annonces De Vente D Appartements A Paris 9eme 75 Seloger Com
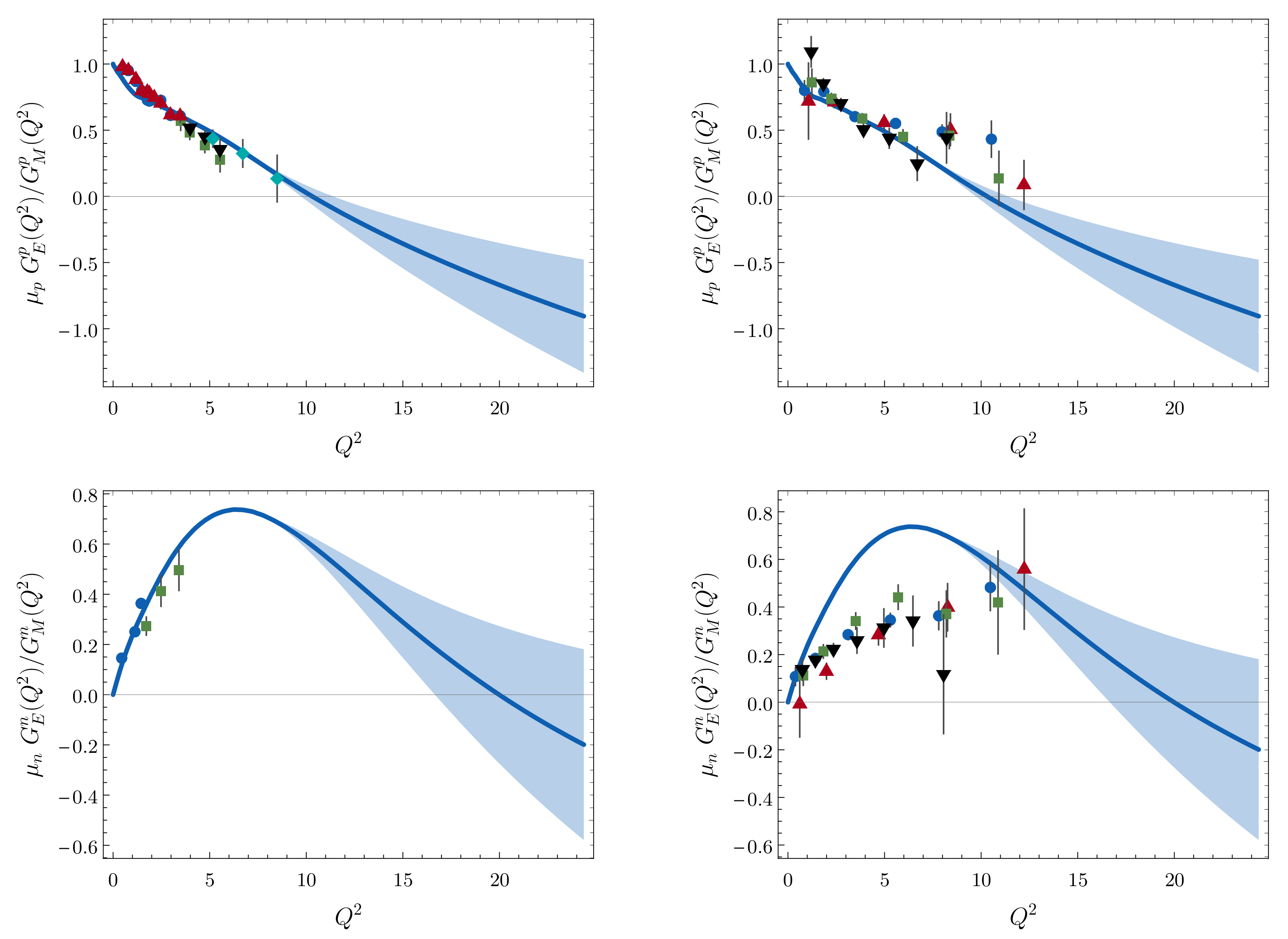



Symmetry Free Full Text Empirical Consequences Of Emergent Mass Html



Images Frandroid Com Wp Content Uploads 21 04 Android 12 Notifications 2 Png



Acp The Impact Of Secondary Ice Production On Arctic Stratocumulus



Hal Archives Ouvertes Fr



Skanaus The Designers Foundry




Quantifying The Contributions Of Tidal Straining And Gravitational Circulation To Residual Circulation In Periodically Stratified Tidal Estuaries In Journal Of Physical Oceanography Volume 40 Issue 6 10



Pubs Rsc Org



Ww2 Ac Poitiers Fr



Sciencedirect Com
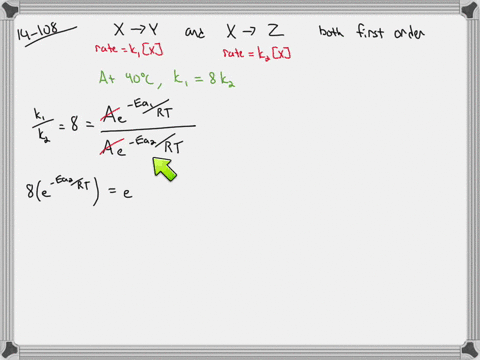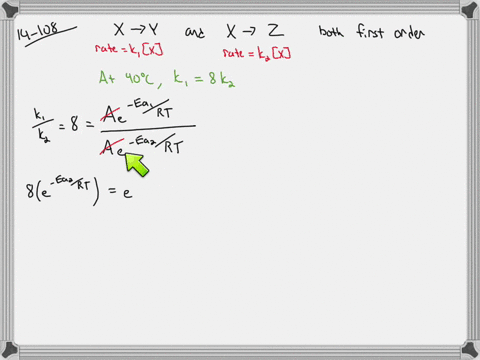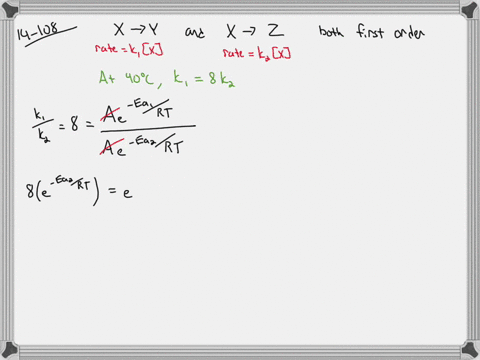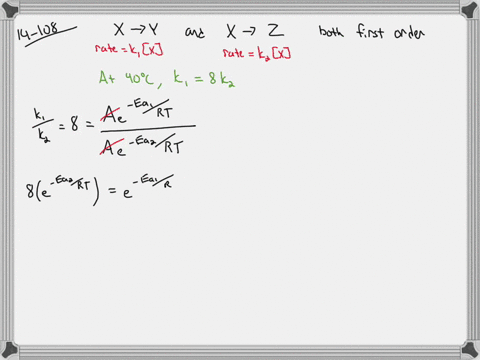



Solved Chlorine Oxide Clo Which Plays An Important Role In The Depletion Of Ozone See Problem 14 106 Decays Rapidly At Room Temperature According To The Equation 2 Mathrm Clo G Longrightarrow Mathrm Cl 2 G Mathrm O 2 G From The




Optimization Of Structural Parameters For Rotary Freestanding Electret Generators And Wind Energy Harvesting Sciencedirect




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr




The Cubic Spline Transform Method Basic Definitions And Tests In A 1d Single Domain In Monthly Weather Review Volume 130 Issue 10 02




Munson Fundamentals Of Fluid Mechanics 5th Chap8 Exercicios Resolvidos Do Capitulo Docsity



Pubs Rsc Org



Mdpi Com




Nyiomb4sfzu7om




Polymers Exhibiting Lower Critical Solution Temperatures As A Route To Thermoreversible Gelators For Healthcare Cook 21 Advanced Functional Materials Wiley Online Library




Utuzwmi Lgahqm




Synthesis Characterization And Photocatalytic Activity Evaluation Of Wo3 Tio2 And Wo3 Tio2 Supported On Zeolite Faujasite



Study And Characterization Of The Micellar Phase Of The Polyethylene Glycol 40 Stearate Water And Soy Lecithin System
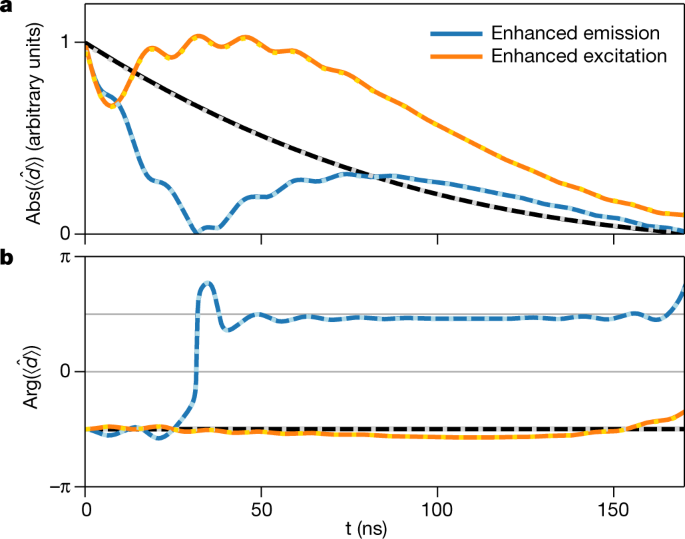



Coherent X Ray Optical Control Of Nuclear Excitons Nature



List Of Unicode Characters Wikipedia



Math Umd Edu




Determining The Radial Distribution Function Of Water Using Electron Scattering A Key To Solution Phase Chemistry The Journal Of Chemical Physics Vol 153 No 19



Ecb Europa Eu



Hal Sorbonne Universite Fr



Orthonormalization Method In Ghost Imaging



Link Aps Org



Jstor Org




Fralda Pom Pom Protea A O De Ma E Protek Tam G 8
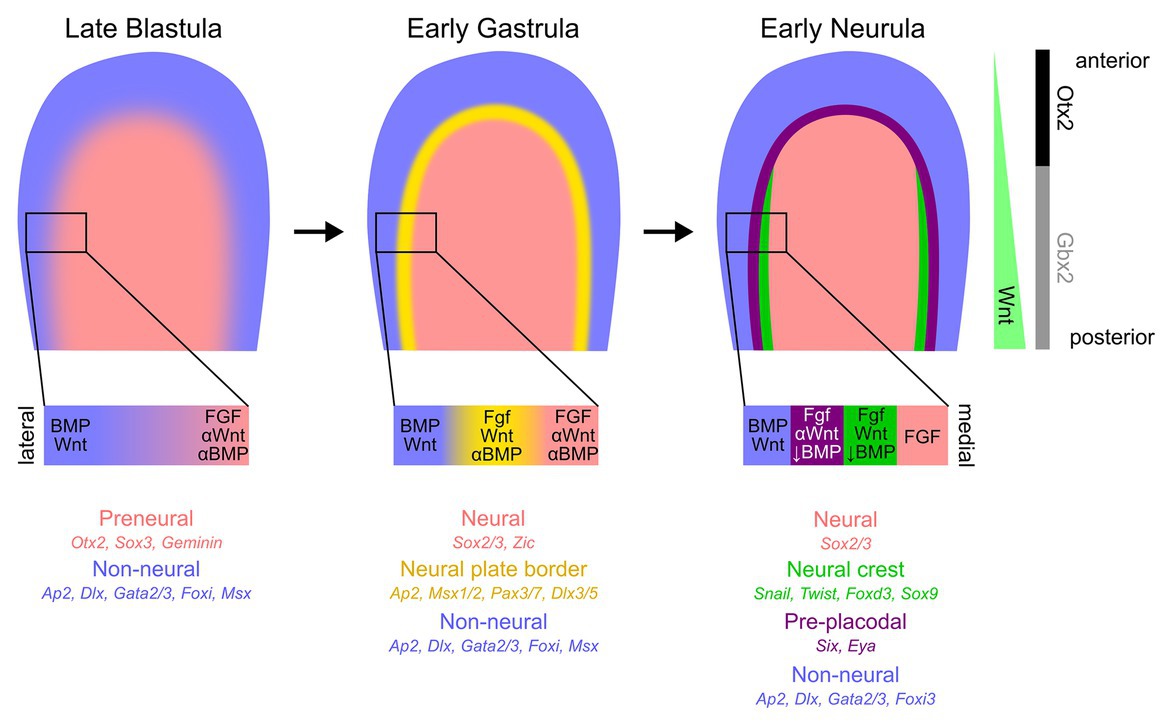



Frontiers Building The Border Development Of The Chordate Neural Plate Border Region And Its Derivatives Physiology




M Asam Magic Finish Fond De Teint Mousse Summer Teint Spf 30 Asambeauty France




Tiles A La Google Maps Coordinates Tile Bounds And Projection Conversion To Epsg 3785 And Epsg 4326 Wgs84 Maptiler




Synthesis Characterization And Photocatalytic Activity Evaluation Of Wo3 Tio2 And Wo3 Tio2 Supported On Zeolite Faujasite



Vector A Has A Magnitude Of 8 00 Units And Makes An Angle Of 45 0 With The Positive X Axis Vector B Also Has A Magnitude Of 8 00 Units And Is Directed Along The



Study And Characterization Of The Micellar Phase Of The Polyethylene Glycol 40 Stearate Water And Soy Lecithin System
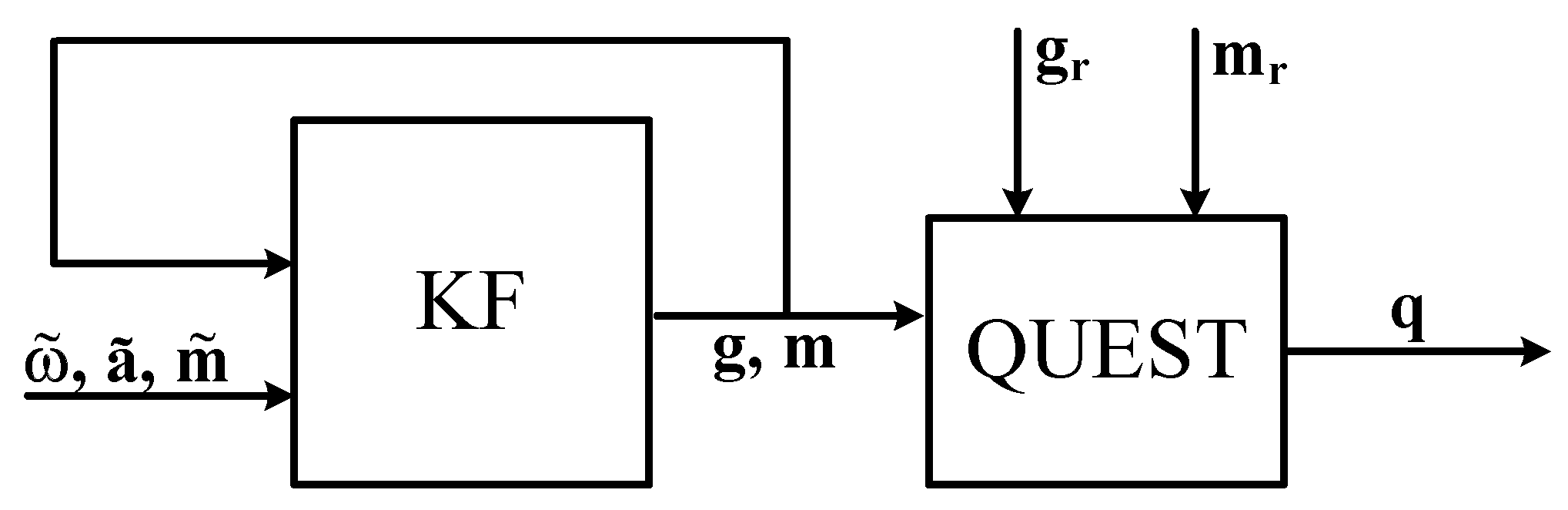



Sensors Free Full Text Survey Of Motion Tracking Methods Based On Inertial Sensors A Focus On Upper Limb Human Motion Html



Study And Characterization Of The Micellar Phase Of The Polyethylene Glycol 40 Stearate Water And Soy Lecithin System



Sujet 19 Groupement Academique 2 Capconcours Cc




A Golf Ball Has A Mass Of 40g And A Speed Of 45m S If The Speed Can Be Measured Within Accuracy Of 2 Calculate The Uncertainty In The Position




Sac Cabas Cuir Noir Le 1440 Medium Noir Matelassage Chevrons Femme Ikks Mode Automne Hiver Sac




2 Resolution Graphique D Equations Du Type F X K Et F X G X Lelivrescolaire Fr




Intervalle Qt 1 Mesure E Cardiogram




A Hierarchical Bayesian Perspective On Majorization Minimization For Non Convex Sparse Regression Application To M Eeg Source Imaging Iopscience



Iopscience Iop Org
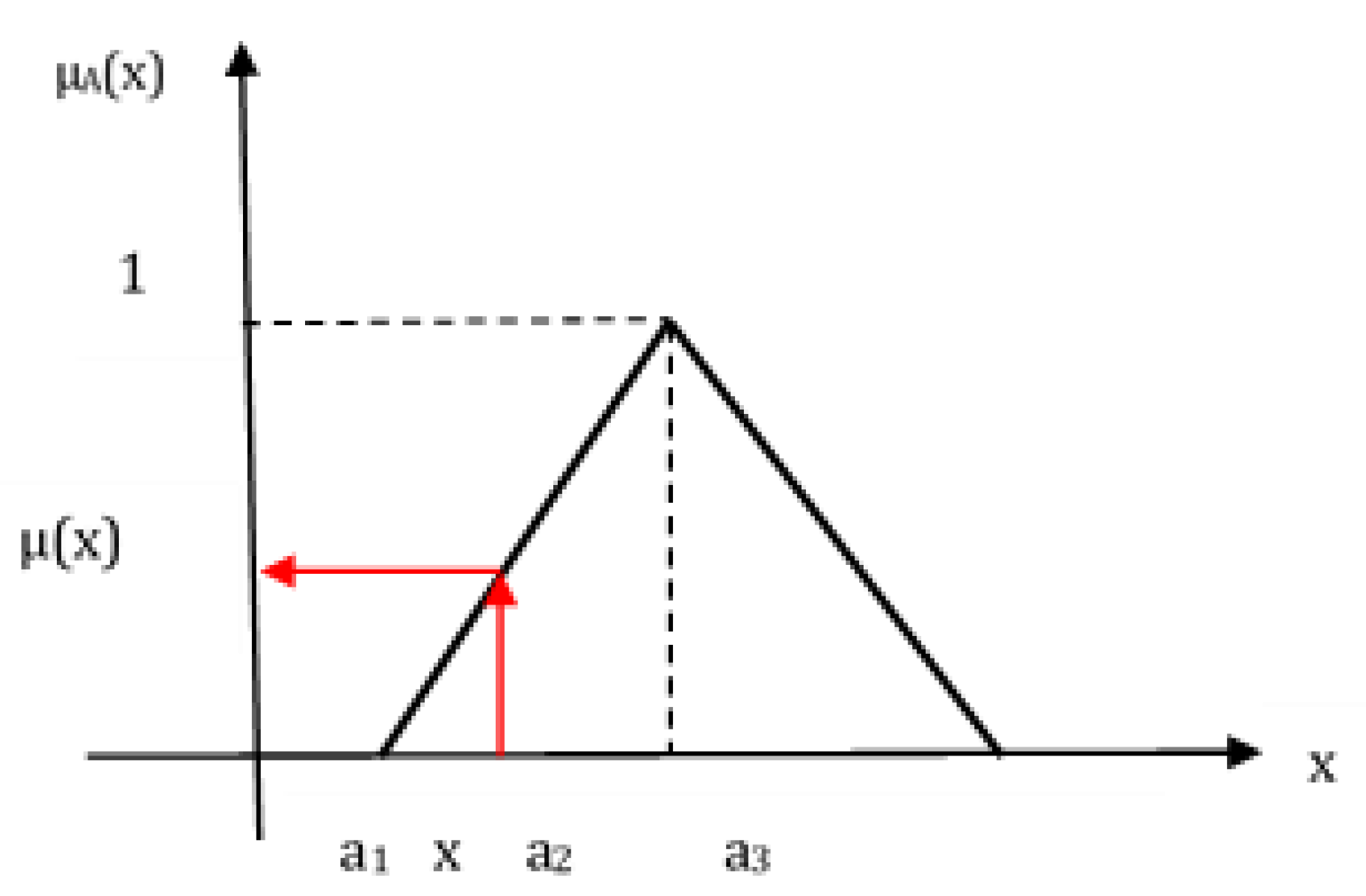



Sustainability Free Full Text A Comparative Analysis Of Fuzzy Topsis And Geographic Information Systems Gis For The Location Selection Of Shopping Malls A Case Study From Turkey Html




Peugeot 404 Wikipedia



Images Frandroid Com Wp Content Uploads 21 04 Android 12 Une Main 2 Jpg



Gi Evaluating The Suitability Of The Consumer Low Cost Parrot Flower Power Soil Moisture Sensor For Scientific Environmental Applications




La Resolution Graphique D Equations Superprof
コメント
コメントを投稿